To understand the complex effects of increased levels of atmospheric carbon dioxide on the oceans, we need to determine where the carbon dioxide from human activity ends up. To begin, we need to calculate the mass of anthropogenic carbon that enters the atmosphere and the resulting change in atmospheric carbon dioxide concentration.
.png)
Worked Example

Atmospheric carbon has important effects on the pH of the oceans. Each year, humans burn about 30 billion barrels of oil and each barrel of oil has a mass of approximately 135 kg. What is the mass of carbon (in kg) that is released when a single barrel of oil is combusted? Assume that the average chemical formula for oil is C8H18(octane) and that complete combustion occurs, producing only carbon dioxide and water.
First, we must write and balance the chemical reaction equation for the combustion of octane:
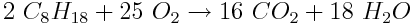
We can use stoichiometry principles to calculate the mass of carbon (in kg) released per 135 kg barrel of oil. First, we will convert the mass of C8H18 into moles. The molar mass of octane is 114.23 g mol-1.
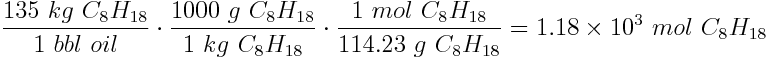
Now we can calculate the mass of carbon released into the atmosphere:
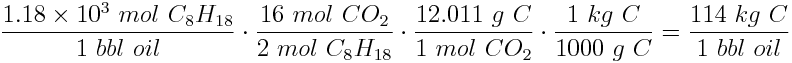
Close to 114 kg of carbon is released into the atmosphere each time a barrel of oil is burned!
Your Turn
Carbon released into the atmosphere in the form of carbon dioxide amplifies atmospheric warming and affects the pH of the ocean. Calculate the amount of carbon, in metric tonnes (1000 kg = 1 tonne), released by human activity each year due to the combustion of approximately 30 billion barrels of oil. Assume that each barrel of oil releases 114 kg of carbon.
One barrel of oil releases 114 kg of carbon. Therefore we can calculate that 30 billion barrels of oil releases:
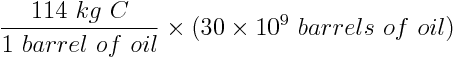
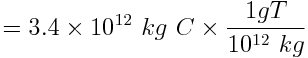
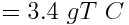
Human combustion of 30 billion barrels of oil releases 3.4 × 1012 kg or 3.4 gigatonnes of carbon into the atmosphere every year.