What Do We Know?
Evidently, as the concentration of aqueous carbon dioxide in the ocean increases, the pH of the ocean decreases. But how does the concentration of dissolved carbon dioxide increase?
Aqueous carbon dioxide is in equilibrium with carbon dioxide in the atmosphere, as follows:
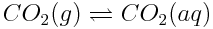
At equilibrium, the concentrations of gaseous carbon dioxide and dissolved carbon dioxide are constant. However, the combustion of fossil fuels, producing more atmospheric carbon dioxide, affects this reaction system in two ways:
- The concentration of a substance in the equilibrium could be changed, causing a change in Q. If the concentrations of substances in a reaction are changed (by adding or removing the substance), then the value of the reaction quotient, Q, changes and Q≠K. If this occurs the reaction will shift, changing the concentrations of reactants and products, until equilibrium is reestablished and Q=K again. If a substance is added to the system, the reaction will shift to remove that substance; if a substance is removed from the system, the reaction will shift to produce more of that substance.
- The temperature of the system could be changed, causing a change in K. Changing the temperature of a chemical system changes the value of K. This explains why equilibrium constants only apply to a reaction when the temperature is specified. Because K changes, Q≠K and the reaction must shift to reestablish equilibrium. If the temperature of the system is raised, the reaction will shift to remove the excess thermal energy; if the temperature of the system is lowered, the reaction will shift to produce more thermal energy.
Note: In reactions involving gases, changing the volume of the reaction mixture causes the concentrations of the substances to change. Therefore, changing the volume changes Q and the reaction shifts until Q=K.